Scientifically reviewed by
Dr. Ky H. Le, MD

The information presented in this article is for educational and research purposes only, intended for laboratory professionals, researchers and collaborators. This content does not constitute medical or clinical advice.
SLU-PP-332 represents a synthetic pan-agonist for estrogen-related receptors with distinct mechanistic properties in metabolic and mitochondrial research.
This compound activates orphan nuclear receptors that govern oxidative metabolism across multiple tissue types. Research applications span skeletal muscle, cardiac tissue, and whole-body energy systems.
The following analysis examines SLU-PP-332’s receptor interactions, transcriptional effects, and experimental outcomes in laboratory models.
Key Highlights
- SLU-PP-332 functions as a pan-ERR agonist with highest selectivity for ERRα, stabilizing active receptor conformations and promoting coactivator recruitment
- The compound drives mitochondrial biogenesis and oxidative metabolism through ERR-PGC-1α transcriptional networks across muscle, cardiac, and metabolic tissues
- In skeletal muscle models, SLU-PP-332 induces oxidative fiber transitions and increases respiratory capacity comparable to exercise-training signatures
- Preclinical studies show systemic metabolic shifts toward fatty acid oxidation, increased energy expenditure, and improved mitochondrial function in stressed tissues
What is SLU-PP-332 Peptide?
SLU-PP-332 is a small molecule compound developed to target estrogen-related receptors.
Unlike classical estrogen receptors, ERRs function as orphan nuclear receptors with no known endogenous ligands. These receptors regulate gene expression programs governing cellular energy metabolism.
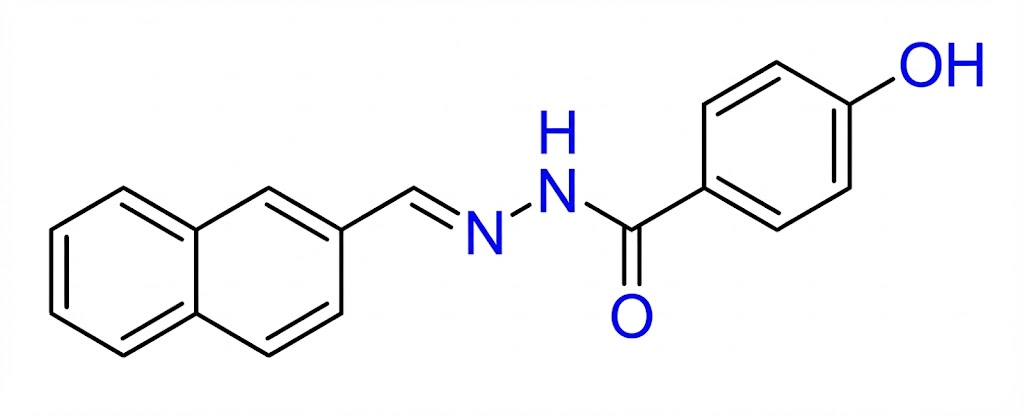
SLU-PP-332 chemical structure
Chemical Classification and Target Profile
The compound acts as a pan-agonist across all three ERR subtypes: ERRα, ERRβ, and ERRγ.
Binding studies demonstrate preferential potency toward ERRα while maintaining activity at ERRβ and ERRγ. This selectivity profile distinguishes SLU-PP-332 from inverse agonists or receptor-selective modulators[1].
Key binding characteristics include:
- Direct interaction with the ERR ligand-binding domain
- Stabilization of active receptor conformations
- Promotion of coactivator recruitment over corepressor binding
- Highest affinity for ERRα compared to ERRβ and ERRγ
ERR Receptor Family Overview
Estrogen-related receptors belong to the nuclear receptor superfamily despite minimal structural homology to classical estrogen receptors.
All three ERR subtypes function as ligand-regulated transcription factors. They bind specific DNA sequences called estrogen-related response elements (ERREs) in target gene promoters[2].
ERR tissue distribution patterns:
- ERRα: High expression in cardiac muscle, skeletal muscle, kidney, and liver
- ERRβ: More restricted distribution with tissue-specific roles
- ERRγ: Limited expression patterns in select metabolic tissues
Together, the three receptors form an integrated network controlling oxidative metabolism.
Related Product: Buy SLU-PP-332 for laboratory research use.
ERR Activation and Transcriptional Control
SLU-PP-332’s mechanism centers on direct receptor binding and downstream transcriptional effects.
The compound functions by altering ERR protein structure in ways that modify gene expression patterns. These changes propagate through multiple metabolic pathways.
Ligand Binding Domain Interactions
The ligand-binding domain of ERRα contains a hydrophobic pocket where SLU-PP-332 docks[1].
Upon binding, the compound induces conformational changes in key receptor regions. The helix-12 domain repositions to create a surface favorable for coactivator protein binding.
This structural rearrangement differentiates agonist from inverse agonist mechanisms. While inverse agonists disrupt coactivator recruitment, SLU-PP-332 enhances it.
Molecular dynamics studies of related ERR ligands show that agonist binding reduces helix-12 flexibility. The stabilized active conformation persists throughout the transcriptional cycle.
PGC-1α Coactivator Recruitment
ERRα forms a critical transcriptional partnership with PGC-1α (peroxisome proliferator-activated receptor γ coactivator-1α).
This coactivator protein functions as a master regulator of mitochondrial biogenesis. PGC-1α itself lacks DNA-binding capacity but amplifies ERR-driven transcription[2].
SLU-PP-332 upregulates PGC-1α-dependent transcriptional programs across multiple tissue types. The ERR-PGC-1α complex activates genes governing oxidative metabolism, mitochondrial function, and fatty acid catabolism.
Without functional PGC-1α, many metabolic effects of ERR activation diminish. This dependence establishes the coactivator as an obligate partner in SLU-PP-332’s mechanism.
The compound essentially acts as a molecular switch. It converts ERR proteins from inactive or weakly active states into robust transcriptional drivers of oxidative gene programs.
Mitochondrial Biogenesis Pathways
Mitochondrial networks respond directly to ERR activation by SLU-PP-332.
These organelles supply ATP through oxidative phosphorylation. Cells adjust mitochondrial content and function based on energy demands.
Nuclear Gene Expression Programs
ERRα, ERRβ, and ERRγ collectively regulate hundreds of genes encoding mitochondrial proteins[2].
SLU-PP-332 upregulates key metabolic gene categories:
- Electron transport chain components: Complex I, III, IV, and V subunits with ERR-responsive promoters
- TCA cycle enzymes: Citrate synthase, isocitrate dehydrogenase, α-ketoglutarate dehydrogenase
- Fatty acid oxidation machinery: CPT1, acyl-CoA synthetase family, β-oxidation enzymes
- Mitochondrial DNA regulation: TFAM and proteins controlling mitochondrial genome transcription
Nuclear-encoded mitochondrial genes show coordinated upregulation across skeletal muscle, cardiac tissue, and metabolic organs following ERR activation.
Respiratory Capacity Measurements
Functional assays confirm that gene expression changes translate into altered mitochondrial performance.
In C2C12 myotubes and cardiac cells, SLU-PP-332 produces measurable functional changes[1]:
- Increased basal and maximal oxygen consumption rates
- Improved respiratory control ratios (ATP-linked respiration to proton leak)
- Enhanced oxidative phosphorylation coupling efficiency
- Normalized TCA intermediate patterns in stressed myocardium (succinate, fumarate, malate)
These functional readouts demonstrate that ERR activation produces metabolically competent mitochondria. The organelles not only increase in number but also operate with improved efficiency.
Laboratory researchers can leverage these effects when studying mitochondrial dysfunction models. SLU-PP-332 provides a pharmacological tool to probe ERR-dependent rescue mechanisms.
Skeletal Muscle Metabolic Remodeling
Skeletal muscle shows particularly robust responses to ERR agonism by SLU-PP-332.
This tissue type exhibits high baseline ERRα expression. Muscle fibers must continuously balance glycolytic and oxidative ATP production based on contraction demands.
Oxidative Fiber Type Transitions
Mammalian skeletal muscle contains multiple fiber types with distinct metabolic profiles.
Fiber type characteristics:
- Type I fibers: Heavy reliance on oxidative metabolism, high fatigue resistance
- Type IIa fibers: Intermediate oxidative capacity
- Type IIx and IIb fibers: Greater glycolytic dependence, lower mitochondrial content
SLU-PP-332 administration shifts fiber composition toward oxidative phenotypes in rodent models. The proportion of type IIa oxidative fibers increases relative to more glycolytic subtypes[3].
This transition requires functional ERRα signaling. Genetic ablation of ERRα prevents the fiber type shift despite continued compound exposure.
The remodeling resembles adaptations observed following endurance exercise training. Both interventions activate similar transcriptional programs through the ERR-PGC-1α axis.
Exercise-Mimetic Gene Signatures
Transcriptomic analysis of SLU-PP-332-treated muscle reveals gene expression patterns matching acute aerobic exercise responses[1].
The compound produces “exercise-mimetic” molecular signatures:
- Upregulation of oxidative phosphorylation components
- Increased fatty acid oxidation enzyme expression
- Enhanced angiogenic factor production
- Activation of genes typically induced by endurance training
Functional testing in mice demonstrates enhanced endurance performance on treadmill protocols. Treated animals run longer distances before exhaustion compared to vehicle controls[3].
This performance improvement depends on ERRα presence. ERRα-knockout animals show no endurance benefit from SLU-PP-332 treatment.
For research applications, SLU-PP-332 enables investigation of ERR-driven metabolic adaptations independent of mechanical or neural factors inherent to actual exercise.
Third-Party Tested, USA-Made, 99% Purity
Lyophilized peptides for laboratory applications. 99% purity, 100% USA-Made.
Cardiac Metabolic Reprogramming
The heart demonstrates distinct metabolic responses to ERR activation.
Cardiac tissue maintains exceptionally high energy demands. The myocardium consumes more ATP per gram than any other organ.
Fatty Acid Oxidation in Myocardium
ERRα and ERRγ serve as central regulators of cardiac energy metabolism under both physiological and stressed conditions[2].
Normal adult myocardium derives 60-90% of ATP from fatty acid oxidation. The remaining energy comes from glucose, lactate, and ketone body oxidation.
SLU-PP-332 and related ERR agonist SLU-PP-915 activate cardiac fatty acid oxidation programs[2]:
- Fatty acid transporters: Upregulation of CD36, fatty acid transport proteins, carnitine shuttle components
- β-oxidation enzymes: Increased expression of long-chain, medium-chain, and short-chain acyl-CoA dehydrogenases
- Metabolite normalization: Restoration of fatty acid profiles toward healthy patterns in failing myocardium
- Acylcarnitine species: Decreased accumulation of long-chain acylcarnitines (markers of incomplete β-oxidation)
Pressure-Overload Model Findings
Cardiac pressure overload models provide experimental systems to study heart failure mechanisms[2].
Transverse aortic constriction (TAC) in mice produces left ventricular hypertrophy and eventual contractile dysfunction. This model recapitulates aspects of hypertensive heart disease.
ERR agonist effects in pressure-overload models[2]:
- Contractile function: Higher left ventricular ejection fraction in treated animals at chronic time points
- Fibrosis reduction: Decreased collagen deposition without necessarily preventing hypertrophy
- Survival benefit: Improved outcomes in some TAC protocols with ERR agonist administration
- Metabolic mechanism: Benefits linked to improved ATP production efficiency rather than blocking hypertrophic response
The failing heart typically shifts away from fatty acid oxidation toward glucose utilization. ERR agonism partially reverses this maladaptive metabolic remodeling.
Laboratory investigation of cardiac metabolism benefits from compounds that probe specific regulatory pathways like ERR signaling.
Systemic Energy Metabolism
SLU-PP-332 produces systemic metabolic effects beyond isolated tissue responses.
Coordinated changes across multiple organs alter whole-organism energy balance. These effects emerge from ERR activation in muscle, adipose tissue, liver, and other metabolic sites.
Substrate Utilization Shifts
Indirect calorimetry measurements in treated animals reveal altered fuel preferences[4].
The respiratory exchange ratio (RER) represents the ratio of CO₂ produced to O₂ consumed. Lower RER values indicate greater fatty acid oxidation relative to carbohydrate usage.
Metabolic shifts observed with SLU-PP-332:
- Decreased RER: Occurs in both lean and diet-induced obese mice, independent of physical activity changes
- Increased fatty acid oxidation: Elevated palmitate oxidation in skeletal muscle, cardiac tissue, and liver
- Improved insulin signaling: Enhanced pathway activation in insulin-resistant models
- Enhanced glucose uptake: Increased glucose transporter expression and uptake capacity in oxidative tissues
These changes support a metabolic state favoring substrate oxidation over storage.
Adipose Tissue Remodeling
White adipose tissue undergoes metabolic reprogramming in response to systemic ERR activation.
Key adipose responses to SLU-PP-332:
- Lipolytic enzyme upregulation: Increased hormone-sensitive lipase and adipose triglyceride lipase
- Oxidative gene expression: Enhanced genes favoring lipid oxidation over storage
- Browning markers: Appearance of thermogenic signatures in some models (UCP1 expression)
- Reduced adiposity: Decreased fat mass accumulation in high-fat diet and genetic obesity models
- Elevated energy expenditure: Primary driver appears to be increased total daily expenditure
These adipose effects complement skeletal muscle and cardiac changes. The multi-tissue coordination produces net reductions in lipid storage with enhanced oxidative disposal.
Laboratory investigations of adipose metabolism and energy balance benefit from tools like SLU-PP-332 that activate defined transcriptional pathways across tissue types.
Research Applications for SLU-PP-332
SLU-PP-332 has multiple potential experimental models in metabolic and mitochondrial research:
| Research Context | Experimental Applications | Relevant Endpoints |
|---|---|---|
| Mitochondrial Biogenesis Studies | Probe ERR-PGC-1α pathway activation; investigate transcriptional control of oxidative genes; examine mitochondrial protein import machinery | Respiratory capacity (OCR), mitochondrial DNA content, TFAM expression, ETC complex activity |
| Skeletal Muscle Metabolism | Model exercise mimetic signaling; study fiber type transitions; investigate oxidative capacity regulation | Fiber typing (immunohistochemistry), gene expression profiling, fatigue resistance assays, metabolomic analysis |
| Cardiac Metabolism Research | Examine fatty acid oxidation pathways; investigate metabolic remodeling in stress models; study ERR-dependent cardioprotection | Metabolite profiling, acylcarnitine analysis, contractile function measurements, fibrosis quantification |
| Whole-Body Energy Balance | Investigate systemic metabolic coordination; examine adipose-muscle crosstalk; study substrate preference regulation | Indirect calorimetry, glucose/insulin tolerance tests, lipidomic profiling |
| Transcriptional Mechanism Studies | Define ERR coactivator dependencies; map ERR target genes; investigate receptor isoform-specific effects | ChIP assays, luciferase reporters, siRNA knockdown experiments, RNA-seq |
| Comparative Pharmacology | Benchmark against other metabolic modulators; study combination effects with PGC-1α activators; examine ERR vs. PPAR pathways | Dose-response curves, pathway activation profiling, gene expression correlation analysis |
Research protocols should account for ERR isoform expression patterns in chosen model systems. ERRα shows broad metabolic tissue distribution, while ERRβ and ERRγ display more restricted expression.
Peptide and small-molecule research compounds require careful quality verification. Triple third-party testing provides confidence in compound identity and purity.
SLU-PP-332 enables mechanistic dissection of ERR-dependent metabolic pathways across multiple physiological contexts. The compound complements other metabolic research tools investigating mitochondrial function and energy metabolism.
Conclusion
SLU-PP-332 functions as a pan-ERR agonist driving oxidative metabolic programs through ERR-PGC-1α transcriptional mechanisms.
The compound produces coordinated effects across metabolic tissues. Mitochondrial biogenesis, enhanced respiratory capacity, and oxidative fiber transitions appear in skeletal muscle models. Cardiac tissue shows metabolic reprogramming with improved fatty acid oxidation in pressure-overload studies.
For research laboratories investigating mitochondrial biology or ERR signaling pathways, SLU-PP-332 provides a defined pharmacological tool to probe metabolic regulatory networks.
BioLongevity Labs provides research-grade SLU-PP-332 with comprehensive analytical verification for in vitro laboratory investigations.
All compounds ship with certificates of analysis from three independent certified laboratories. USA-based GMP manufacturing and rigorous third-party testing ensure consistent quality for reproducible research outcomes.
Products are strictly for research purposes only and not for human consumption.
Scientific Reviewer
This research article has been scientifically reviewed and fact-checked by Dr. Ky H. Le, MD. Dr. Le earned his medical degree from St. George’s University School of Medicine and completed his residency training at Memorial Hermann Southwest Hospital. Board-certified in family medicine with experience in hospital medicine, he brings over two decades of clinical experience to reviewing research content and ensuring scientific accuracy.
About BioLongevity Labs
BioLongevity Labs manufactures research-grade peptides in USA GMP facilities with triple third-party testing verification. Every batch includes comprehensive analytical documentation and COAs. Browse our lyophilized peptide vials to find compounds for your laboratory applications.
References
- Billon C, Sitaula S, Banerjee S, Welch R, Elgendy B, Hegazy L, et al. Synthetic ERRα/β/γ Agonist Induces an ERRα-Dependent Acute Aerobic Exercise Response and Enhances Exercise Capacity. American Chemical Society (ACS); 2023. https://doi.org/10.1021/acschembio.2c00720
- Xu W, Billon C, Li H, Wilderman A, Qi L, Graves A, et al. Novel Pan-ERR Agonists Ameliorate Heart Failure Through Enhancing Cardiac Fatty Acid Metabolism and Mitochondrial Function. Ovid Technologies (Wolters Kluwer Health); 2024. https://doi.org/10.1161/circulationaha.123.066542
- Nasri H. New hopes on “SLU-PP-332” as an effective agent for weight loss with indirect kidney protection efficacy; a nephrology point of view. Maad Rayan Publishing Company; 2024. https://doi.org/10.34172/jre.2024.25143
- Billon C, Schoepke E, Avdagic A, Chatterjee A, Butler AA, Elgendy B, et al. A Synthetic ERR Agonist Alleviates Metabolic Syndrome. Elsevier BV; 2024. https://doi.org/10.1124/jpet.123.001733
