The information presented in this article is for educational and research purposes only, intended for laboratory professionals, researchers and collaborators. This content does not constitute medical or clinical advice.
Understanding peptide nomenclature is essential for any research lab working with these important biological molecules. Peptides are chains of amino acids linked together by peptide bonds, forming everything from simple dipeptides (two amino acids) to complex polypeptide structures.
Whether you’re conducting in vitro studies, developing new assays, or sourcing peptides for your experiments, knowing how to properly name peptides ensures clear communication and accurate results.
This comprehensive guide covers the official IUPAC-IUBMB naming system that scientists worldwide use to identify amino acids and peptides. We’ll break down everything from basic amino acid codes to complex modification nomenclature in simple, practical terms.
Why Peptide Nomenclature Matters for Research
Proper nomenclature prevents costly mistakes in research. When ordering peptides from suppliers, incorrect naming can lead to receiving the wrong compound entirely. Standard naming systems also ensure your published research is clearly understood by the global scientific community.
The International Union of Pure and Applied Chemistry (IUPAC) and International Union of Biochemistry and Molecular Biology (IUBMB) established these guidelines to create universal consistency. Every researcher, from graduate students to senior investigators, benefits from using the same naming system.
The 20 Standard Amino Acids
Basic Amino Acid Codes
Every peptide is built from amino acid building blocks. The 20 standard amino acids each have unique three-letter and one-letter codes that form the foundation of peptide nomenclature.
| Amino Acid | 3-Letter | 1-Letter | Chemical Class |
|---|---|---|---|
| Alanine | Ala | A | Non-polar |
| Arginine | Arg | R | Basic |
| Asparagine | Asn | N | Polar |
| Aspartic acid | Asp | D | Acidic |
| Cysteine | Cys | C | Polar |
| Glutamine | Gln | Q | Polar |
| Glutamic acid | Glu | E | Acidic |
| Glycine | Gly | G | Non-polar |
| Histidine | His | H | Basic |
| Isoleucine | Ile | I | Non-polar |
| Leucine | Leu | L | Non-polar |
| Lysine | Lys | K | Basic |
| Methionine | Met | M | Non-polar |
| Phenylalanine | Phe | F | Non-polar |
| Proline | Pro | P | Non-polar |
| Serine | Ser | S | Polar |
| Threonine | Thr | T | Polar |
| Tryptophan | Trp | W | Non-polar |
| Tyrosine | Tyr | Y | Polar |
| Valine | Val | V | Non-polar |
Chemical Classification Breakdown
Understanding amino acid properties helps predict peptide behavior in experiments. Non-polar amino acids make up 45% of the standard set, while polar amino acids account for 30%. Basic amino acids comprise 15%, and acidic amino acids represent 10%.
These proportions reflect the diverse chemical environments peptides encounter in biological systems. When designing experiments, consider how these different chemical classes might affect peptide solubility, stability, and interactions.
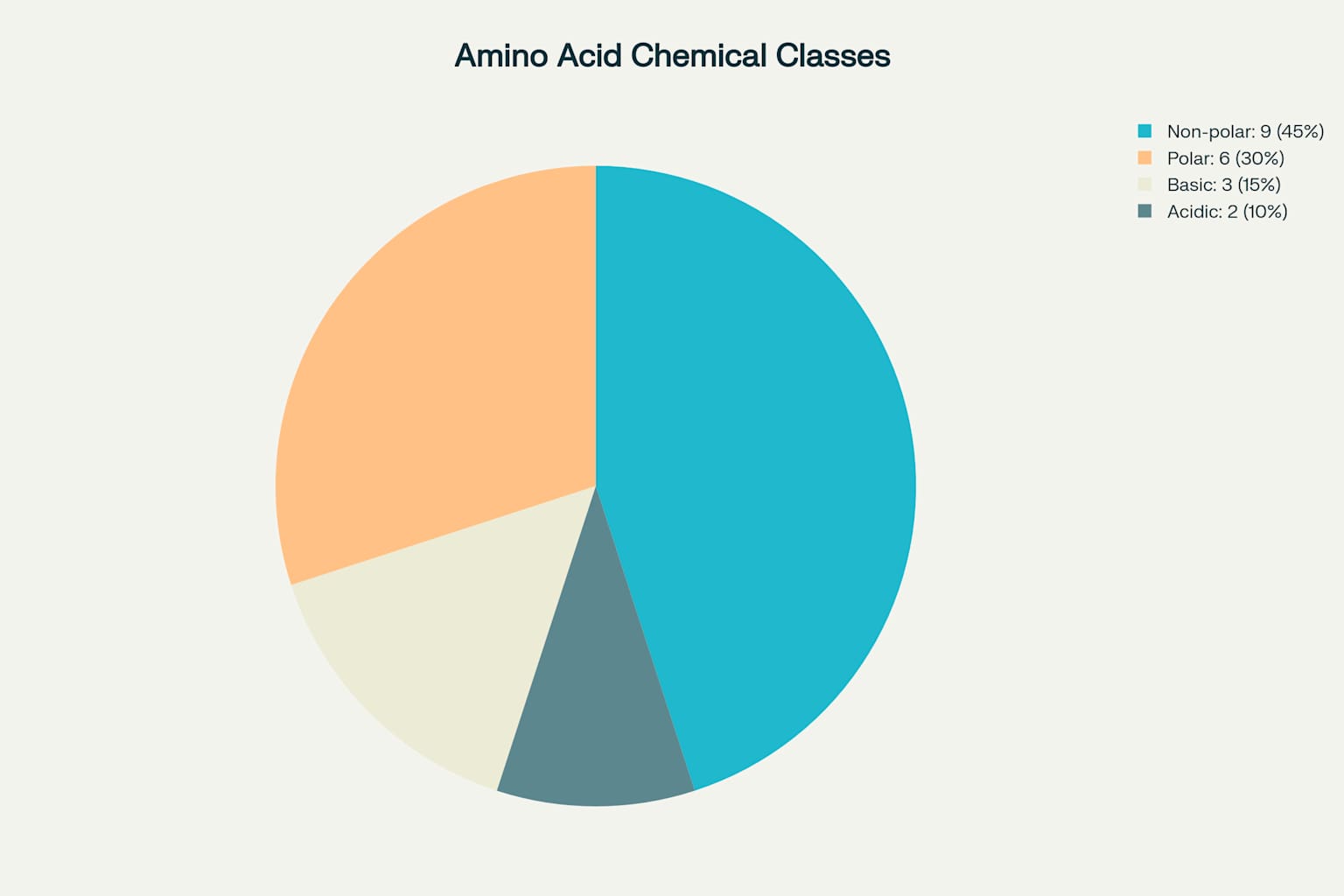
Distribution of chemical classes among the 20 standard amino acids used in protein synthesis
Extended and Special Amino Acid Codes
Beyond the Standard 20
Research often involves amino acids beyond the basic 20. The nomenclature system includes codes for these special cases to maintain clarity in complex studies.
| Amino Acid | 3-Letter | 1-Letter | Description |
|---|---|---|---|
| Selenocysteine | Sec | U | 21st amino acid |
| Pyrrolysine | Pyl | O | 22nd amino acid |
| Aspartic acid or Asparagine | Asx | B | Used when uncertain |
| Glutamic acid or Glutamine | Glx | Z | Used when uncertain |
| Any amino acid | Xaa | X | Universal placeholder |
Selenocysteine and pyrrolysine are incorporated into proteins through specialized mechanisms. The ambiguous codes (B and Z) prove especially useful when mass spectrometry cannot distinguish between closely related amino acids during protein sequencing.
Non-Standard Amino Acids in Research
Common Non-Standard Amino Acids
Synthetic peptides and specialized proteins often contain modified amino acids. These require specific three-letter codes for accurate identification and ordering.
Key Non-Standard Amino Acids:
- Abu (2-Aminobutanoic acid) – Shorter chain analog of valine
- Aib (α-Aminoisobutyric acid) – α-methylalanine, increases peptide stability
- Ahx (6-Aminohexanoic acid) – Commonly used as a linker in peptide synthesis
- Cit (Citrulline) – Found in the urea cycle and immune responses
- Orn (Ornithine) – Lysine analog important in metabolic pathways
- Hyp (Hydroxyproline) – Most abundant in collagen
Modified Amino Acids
Post-translational modifications create additional amino acid variants that appear frequently in research applications.
Common Modifications:
- pSer, pThr, pTyr – Phosphorylated amino acids
- Glp or pGlu – Pyroglutamic acid (cyclized glutamic acid)
- Gla – γ-Carboxyglutamic acid (vitamin K-dependent modification)
- MeLeu, MePhe – N-methylated amino acids
Peptide Naming Rules
The Basic System
Peptide names follow strict rules that prevent confusion. Always write sequences from the N-terminal (amino) end to the C-terminal (carboxyl group) end, reading left to right. This convention mirrors the direction of protein synthesis in cells.
For systematic naming, all amino acids except the C-terminal residue use their acyl forms (ending in -yl). The C-terminal residue keeps its full amino acid name. Each peptide bond creates an amide linkage between the carboxyl group of one amino acid and the amino group of another. For example, Gly-Ala becomes glycylalanine.
Two Abbreviation Systems
Three-Letter System:
- Best for sequences up to 10-20 residues
- Residues connected with hyphens: His-Phe-Arg-Trp-Pro
- First letter capitalized, others lowercase
- Clear and readable for shorter peptides, from simple tripeptides to longer peptide chains
One-Letter System:
- Used for longer sequences and proteins
- No separators needed: HFRWP
- More compact for database storage
- Essential for bioinformatics applications and polymer analysis
Modifications and Special Notations
Common Modification Types
Research peptides frequently contain modifications that alter their properties. Proper notation ensures accurate communication about these important structural features.
| Modification | Example | Description |
|---|---|---|
| Cyclic peptides | cyclo(Gly-Ala-Leu) | Head-to-tail cyclization |
| Disulfide bonds | Cys³-Cys⁸ | Numbers show linked positions |
| N-terminal modification | Ac-Gly-Ala | Ac = acetyl group |
| C-terminal modification | Gly-Ala-NH₂ | Amidated C-terminus |
| Residue replacement | [Ala⁵]peptide | Alanine substitution at position 5 |
Notation Symbols
Understanding these symbols helps interpret complex peptide structures:
- Brackets [ ] indicate modifications or substitutions
- Parentheses ( ) show connectivity or protection groups
- Superscript numbers specify residue positions
- Prefixes like D- indicate stereochemical configuration
Real-World Examples
Enkephalins: Classic Research Peptides
The enkephalins demonstrate proper nomenclature in action. These endogenous opioid peptides are frequently used in pain research and receptor studies.
Met-enkephalin:
- Three-letter: Tyr-Gly-Gly-Phe-Met
- One-letter: YGGFM
- Systematic name: Tyrosylglycylglycylphenylalanylmethionine
Leu-enkephalin:
- Three-letter: Tyr-Gly-Gly-Phe-Leu
- One-letter: YGGFL
- Systematic name: Tyrosylglycylglycylphenylalanylleucine
Hormone Peptides
Peptide hormones showcase the nomenclature system’s application to complex biological molecules commonly studied in endocrinology research.
Oxytocin:
- Sequence: Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH₂
- Type: Cyclic nonapeptide with disulfide bridge
- Key features: Amidated C-terminus, internal disulfide bond
Working with Suppliers
Ordering Best Practices
When sourcing peptides for research, proper nomenclature prevents costly errors. Always provide both three-letter and one-letter sequences when possible. Specify any modifications clearly using standard notation.
Include stereochemical information when relevant. If your peptide contains D-amino acids, unusual modifications, or specific cyclization patterns, use the proper notation to ensure accurate synthesis.
Quality Control Considerations
Reputable peptide suppliers use standard nomenclature in their documentation. Peptide quality certificates should list peptides using proper IUPAC-IUBMB naming conventions. This consistency helps verify that you received the correct compound for your experiments.
Stereochemistry and Configuration
L and D Amino Acids
Most naturally occurring amino acids exist in the L-configuration. However, research peptides may contain D-amino acids for increased stability or altered biological activity. The nomenclature system accommodates these variations.
Examples:
- L-Ala-L-Leu (can be written as Ala-Leu when all residues are L)
- Gly-D-Ala-Leu (specifically indicates D-alanine at position 2)
- D-Phe-cyclo(Cys-Tyr-D-Trp-Lys-Thr-Cys)-Thr-NH₂ (complex example with multiple D-residues)
Advanced Nomenclature Features
Ionization States
Peptides exist in different ionization states depending on pH conditions. The nomenclature acknowledges these forms, which affect peptide behavior in biological systems and experimental conditions.
At physiological pH, most peptides exist as zwitterions with both positive and negative charges. Understanding these states helps predict peptide behavior in cell culture media and buffer systems.
Residue Numbering
Peptide positions are numbered from the N-terminal end starting with position 1. This system enables precise communication about specific modifications, substitutions, or binding sites within peptide sequences.
Position numbering becomes crucial when discussing structure-activity relationships or comparing peptide variants in research publications.
Summary
Mastering peptide nomenclature enhances research accuracy and communication. Whether you’re designing new experiments, publishing results, or sourcing compounds from suppliers, proper naming conventions ensure clarity and prevent errors.
The IUPAC-IUBMB system provides a robust framework that accommodates everything from simple dipeptides to complex modified sequences. By following these established guidelines, researchers contribute to the clear, consistent communication that advances peptide science.
Remember that proper nomenclature is not just about following rules. It’s about ensuring your research can be understood, reproduced, and built upon by scientists worldwide. When in doubt, refer to official IUPAC-IUBMB guidelines or consult with experienced peptide suppliers who understand these naming conventions.
