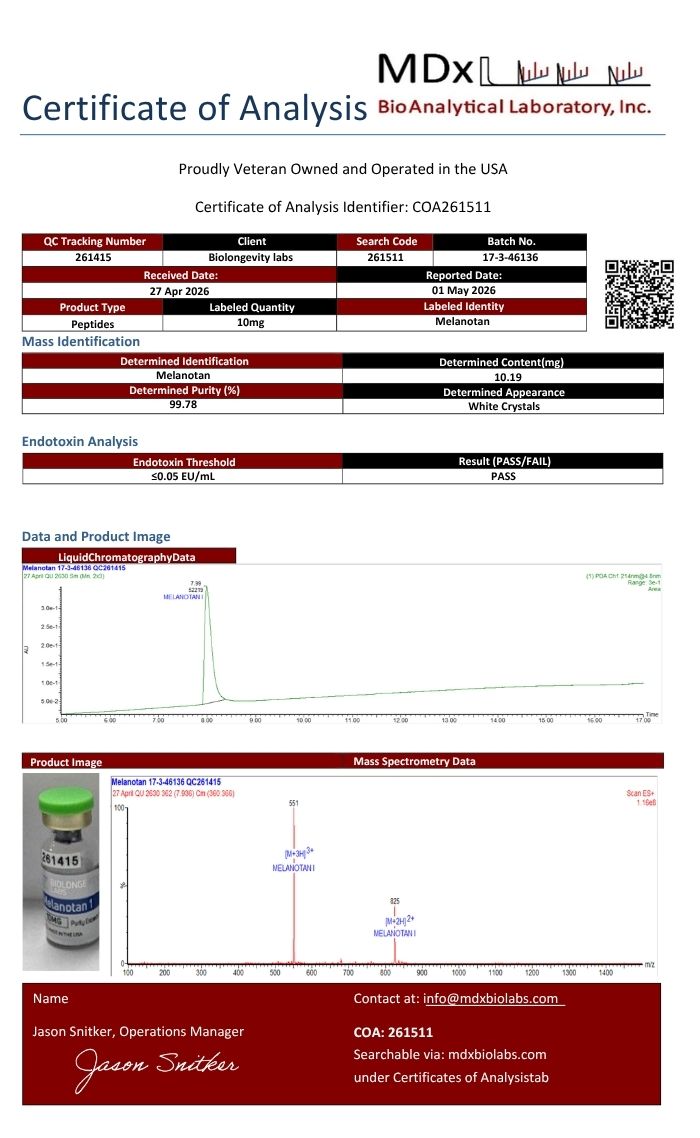
Melanotan 1 Description
Melanotan-1 (afamelanotide, melanotan-I) is a synthetic analog of alpha-melanocyte stimulating hormone (α-MSH). This peptide hormone analog demonstrates activity at melanocortin receptors in laboratory studies.
Research indicates that Melanotan-1 binds to melanocortin-1 receptors (MC1R), stimulating melanogenesis pathways in melanocyte cell cultures. This receptor activation triggers melanin synthesis through the cyclic AMP signaling cascade. Studies show the compound produces melanin without requiring UV exposure in in vitro melanocyte models.
This peptide is provided strictly for in vitro research purposes in qualified laboratory settings. Researchers studying melanocortin biology and pigmentation mechanisms use Melanotan-1 in controlled experimental protocols for investigating cellular signaling pathways.
Melanotan-1 serves research laboratories studying:
- Melanocortin receptor signaling pathways
- Melanogenesis mechanisms in cell culture systems
- Photoprotection pathways in dermal cell models
- α-MSH analog interactions with MC1R
- Pigmentation biology in controlled laboratory settings
Peptide Information
| Property |
Value |
| Peptide Sequence |
Ser-Tyr-Ser-Nle-Glu-His-D-Phe-Arg-Trp-Gly-Lys-Pro-Val |
| Molecular Formula |
C78H111N21O19 |
| Molecular Weight |
1646.8 g/mol |
| CAS Number |
75921-69-6 |
| PubChem CID |
16154396 |
| Synonyms |
Melanotan I, Melanotan-1, 75921-69-6, NDP-alpha-MSH, 4-Nle-7-phe-alpha-msh, MT1 |
Lyophilized Peptides:
These peptides are freeze-dried, a process that not only extends shelf life but also preserves the purity and integrity of the peptides during storage. We do not use any fillers in this process.
Melanotan-1 Research Overview
Melanotan-1 is a synthetic analog of alpha-melanocyte stimulating hormone (α-MSH). The peptide binds to melanocortin receptors, particularly MC1R, through a conserved sequence containing histidine, phenylalanine, arginine, and tryptophan residues[1].
The compound shows enhanced stability compared to endogenous α-MSH. Research examines its effects across multiple physiological systems where melanocortin signaling operates.
Melanogenesis Research
MC1R binding initiates cAMP-dependent signaling cascades in melanocytes. This activation upregulates tyrosinase and related enzymes that control pigment synthesis[2].
The signaling pathway promotes eumelanin production rather than pheomelanin. Expression of microphthalmia-associated transcription factor increases, regulating downstream pigmentation genes[3].
DNA Repair Mechanisms
MC1R activation enhances nucleotide excision repair through cAMP-dependent pathways. Protein kinase A phosphorylates ATR at serine 435, improving XPA recruitment to DNA damage sites[4].
The pathway accelerates removal of UV-induced photolesions. Base excision repair enzymes like OGG1 and APE1 show increased expression.
Anti-Inflammatory Pathways
Melanocortin receptor engagement on macrophages modulates NF-κB signaling. MC1R and MC3R activation reduces pro-inflammatory cytokine production and nitric oxide generation[5].
Anti-inflammatory mediator production increases, including IL-10. In neural injury models, microglial activation decreases and CD206-expressing phenotypes emerge[5][6].
Leukocyte adhesion and neutrophil infiltration decline in affected tissues[7].
Oxidative Stress Response
MC1R signaling modulates reactive oxygen species production and antioxidant defenses. Expression of superoxide dismutase, glutathione peroxidase, and catalase increases[8].
The pathway activates PI3K/Akt/Nrf2 signaling in neural tissue. Phase II detoxification enzymes and antioxidant proteins show enhanced expression[8].
Mitochondrial dynamics shift, affecting fission-fusion balance and uncoupling protein expression[9].
Neuroprotective Studies
MC1R activation influences AMPK, SIRT1, and PGC-1α pathways that regulate mitochondrial metabolism. Oxidative phosphorylation efficiency and ATP production change[8].
Apoptotic signaling shifts toward anti-apoptotic proteins. Oxidative damage markers decrease in cerebral injury models. Caspase-mediated death pathways show reduced activation[6][8].
Blood-brain barrier integrity parameters shift in experimental systems[10].
Hepatic Research
Melanocortin signaling reduces hepatic neutrophil infiltration in endotoxin models. Systemic nitric oxide production decreases[11].
Cytokine and chemokine expression patterns change in liver tissue[12]. Cellular stress response pathways may be involved, though mechanisms need more study.
Erythropoietic Research
Studies in erythropoietic protoporphyria suggest effects on erythroblast survival. Observations indicate potential modulation of protoporphyrin accumulation[13].
Molecular mechanisms in erythroid cell populations require additional research. Melanocortin receptor signaling pathways known in other cell types may apply.
References
- A. Polańska et al., “Afamelanotide in protoporphyria and other skin diseases: a review,” Termedia Sp. z.o.o., 2024. doi: 10.5114/ada.2024.138818.
- F. Wang, W. Ma, D. Fan, J. Hu, X. An, and Z. Wang, “The biochemistry of melanogenesis: an insight into the function and mechanism of melanogenesis-related proteins,” Frontiers Media SA, Aug. 2024. doi: 10.3389/fmolb.2024.1440187.
- S. Ma et al., “Structural mechanism of calcium-mediated hormone recognition and Gβ interaction by the human melanocortin-1 receptor,” Springer Science and Business Media LLC, Aug. 2021. doi: 10.1038/s41422-021-00557-y.
- E. M. Wolf Horrell, M. C. Boulanger, and J. A. D’Orazio, “Melanocortin 1 Receptor: Structure, Function, and Regulation,” Frontiers Media SA, May 2016. doi: 10.3389/fgene.2016.00095.
- H. B. Patel, T. Montero-Melendez, K. V. Greco, and M. Perretti, “Melanocortin Receptors as Novel Effectors of Macrophage Responses in Inflammation,” Frontiers Media SA, 2011. doi: 10.3389/fimmu.2011.00041.
- X. Wu et al., “NDP-MSH binding melanocortin-1 receptor ameliorates neuroinflammation and BBB disruption through CREB/Nr4a1/NF-κB pathway after intracerebral hemorrhage in mice,” Springer Science and Business Media LLC, Oct. 2019. doi: 10.1186/s12974-019-1591-4.
- W. Xu et al., “Activation of Melanocortin 1 Receptor Attenuates Early Brain Injury in a Rat Model of Subarachnoid Hemorrhage viathe Suppression of Neuroinflammation through AMPK/TBK1/NF-κB Pathway in Rats,” Elsevier BV, Jan. 2020. doi: 10.1007/s13311-019-00772-x.
- W. Xu et al., “Melanocortin 1 receptor attenuates early brain injury following subarachnoid hemorrhage by controlling mitochondrial metabolism via AMPK/SIRT1/PGC-1α pathway in rats,” Ivyspring International Publisher, 2021. doi: 10.7150/thno.49426.
- S. Fu et al., “Activation of the Melanocortin-1 Receptor by NDP-MSH Attenuates Oxidative Stress and Neuronal Apoptosis through PI3K/Akt/Nrf2 Pathway after Intracerebral Hemorrhage in Mice,” Wiley, Nov. 2020. doi: 10.1155/2020/8864100.
- S. Yu et al., “Activation of MC1R with BMS-470539 attenuates neuroinflammation via cAMP/PKA/Nurr1 pathway after neonatal hypoxic-ischemic brain injury in rats,” Springer Science and Business Media LLC, Jan. 2021. doi: 10.1186/s12974-021-02078-2.
- A.-E. Minder et al., “Beyond pigmentation: signs of liver protection during afamelanotide treatment in Swiss patients with erythropoietic protoporphyria, an observational study,” SAGE Publications, Jan. 2021. doi: 10.1177/26330040211065453.
- H. Chiao, S. Foster, R. Thomas, J. Lipton, and R. A. Star, “Alpha-melanocyte-stimulating hormone reduces endotoxin-induced liver inflammation.,” American Society for Clinical Investigation, May 1996. doi: 10.1172/jci118639.
- J. G. Langendonk et al., “Afamelanotide for Erythropoietic Protoporphyria,” Massachusetts Medical Society, Jul. 2015. doi: 10.1056/nejmoa1411481.